The tech that could help us fight diabetes
There are several technologies in development, but the message is "don't get your hopes up"
- As it's the start of Diabetes Week in the UK, we're taking a look at the technological advances around right now to help those fighting the disease - and whether help is really just over the horizon.
The funny thing about today's health gadgets is that many of them are aimed at relatively healthy people – people for whom the biggest lifestyle challenge is fitting into a pair of jeans from 2012, or doing a predefined activity faster than last week so that people on Facebook 'like' it.
These devices can be little more than mechanical vanity hands that continually pat the wearer on the back, or clap him or her to a new PB. Fitbit is great for people who are already a bit fit.
Some people, though, aren't as fortunate. Managing Type 1 diabetes (and insulin-dependent Type 2) is a bit of a crude body hack at the best of times.
People used to always die because of T1, now they don't. But, while this is a miracle of modern medicine, people who live with Type 1 can be forgiven for not shouting the good news from the rooftops when their daily routine is governed by blood tests, and injections or insulin pumps, in order to maintain a healthy equilibrium.
What sufferers want is for technology to provide a wireless, needle-free way of checking blood glucose. Such an innovation has been promised for 30 years, but while it seems like it should be relatively easy to achieve it's yet to become a reality – it's forever just over the horizon, and that's where it's stayed.
So what's holding this miracle tech back?
The problem
The food we eat is converted to glucose for fuel, but in Type 1 diabetics that glucose stays in the bloodstream because their body doesn't produce the insulin needed to store it away; this is a job the body does automatically, and really well, in people without diabetes.
Get daily insight, inspiration and deals in your inbox
Sign up for breaking news, reviews, opinion, top tech deals, and more.

So diabetics inject or pump insulin into themselves manually to get the glucose out of the blood, hence the need for numerous daily blood tests to make sure you're not too high (long-term bad, damage to blood vessels) or too low (short-term bad, fainting, falling over, crashing the car).
The key to all this is knowing your current blood glucose level, and the best way of doing this at the moment is to literally put some blood on a plastic strip and see what it says – there's no sci-fi eyeball/skin/toe scanner that can do this job.
It's a weird necessity, and one you'd have thought today's technology would've long ago solved via a $699 gadget you stick your finger in or tape to your stomach and forget about, only removing it once a month to recalibrate it by blowing out the fluff. But no.
When you're living with daily blood tests to check glucose, every tech announcement is a disappointment. Some thought the Apple Watch might monitor glucose, but of course all it does is present health data from other sources in a slightly jazzier way.
The frustrating thing is that the problem seems like it should be solvable. How come they can do so much on 'House' to scan patients' bodies, but there's still no real-world health gadget that can check glucose without needing blood to be drawn by the modern equivalent of a leech?
Yes, there are some continuous glucose monitors (CGMs) out there right now, but even these require a wire to be poked into the skin, a patchy solution at best. Plus they're expensive, so good luck getting your local health provider to agree to handing one over.
Sadly, there's currently no way to measure how much glucose is in the blood without poking something into the body, like a crude dipstick, and seeing what it says.
Unless a few companies deliver on their promises, that is...
The best of today
The best option we have at the moment is established medical firm Abbott's Freestyle Libre, a wireless device that attaches to the wearer's skin and takes a continuous measurement of glucose which is analyzed via a tap from a reader.
Yes, a proper health wearable. The problem is that it tests by dangling a small needle into the skin (a microscopic hair-like affair, so there's no actual stab), so it needs to be firmly attached.
It also only tests the interstitial fluid (the transparent liquid that appears along with blood when you cut yourself), which is less quickly refreshed by the body, so the data it generates can be a little old.
Simon O'Neill from Diabetes UK says of this method: "CGMs are good, but they measure the interstitial fluid, leading to a time lag of 15 to 20 minutes, so they may say you're fine when you're not."
In fact, in 20 minutes the body can go from running as normal to crashing lows when the insulin is really packing away the glucose, so this method is a bit of a rough fix at best.
That said, it gives a slightly time-shifted but nevertheless accurate graph of blood glucose, an extremely useful thing to have if you're trying to continually balance food intake to insulin.
Steve Scott, divisional VP of technical operations at Freestyle maker Abbott, is understandably happy with the tech, telling us: "...being able to remove that finger-prick for people to be able to manage their glucose levels, while also empowering both them and their healthcare providers to improve their glycemic control and ultimately achieve better health, has been a game changer".
Indeed, any online diabetes forum is now packed with people sharing their Libre graphs, showing that, for super-engaged diabetics at least, having a live stream of data is definitely helping them get to grips with the condition. Sadly, the sensors are hardwired to last for only 14 days, and they're £50 a pop.
Explaining how the Libre came about tells you why the technology is a long way from being a menu option on a smartwatch. Scott adds "...over the last few years we have been able to progress the chemistry to be stable enough for the 14-day wear time, while also remove the need for calibration," which, although seemingly quite a small leap to the average user, is actually a huge technical jump.
For some, though, the constant data this generates can be a nightmare. "They call it alarm fatigue," said Simon O'Neill, healthcare intelligence director at Diabetes UK. "People who aren't so engaged with staying on top of their diabetes end up with a device that's only telling them they're failing all the time, so they switch it off."
Which is one of the worst things about Type 1 diabetes. It never stops. You can't take a break.
The science of tomorrow
What diabetes sufferers would really, really like is a noninvasive glucose tester. Something that can test the blood sugar level without needing a needle or a poke, making it less of a weird thing to be seen doing while also giving a live reading to fend off the ever present horror of the 'hypo' – a glucose level so low that the sufferer goes wobbly or crashes out.
Something like the GlucoWise, one of 2016's new wave of potential non-invasive testers, and the latest concept device to promise a full noninvasive test by beaming something through any loose bits of skin you may have. It's a proper Star Trek solution, the fuss-free scanner of our dreams – if it turns out to be more than a pipe dream.
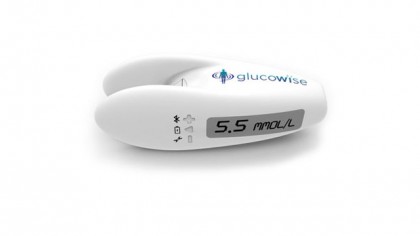
"This is our third revision," Dr George Palikaras, president and CEO of GlucoWise's parent company MediWise, says of the impressive renders of the GlucoWise sensor that are currently exciting diabetics, who are hoping this isn't simply the latest form of vaporware to hit the noninvasive testing world.
"Versions 1 and 2 were on the earlobe, but the feedback from focus groups was that people wanted discretion," he said, adding: "We cannot penetrate bone, hence you can use it on the lip, eyebrow... anywhere that's soft tissue."
"We've been working on this for four and a half years," he adds, telling us more than a few times that there's still quite a long way to go before GlucoWise becomes an actual product.
Palikaras explains that the root of the problem is our skin. "The body is a harsh environment," he says. "Skin does a great job of protecting it, reflecting 90 to 95 per cent of energy you put at it."
But how does GlucoWise manage to penetrate skin, even the floppy bit between thumb and forefinger its renders suggest using? Palikaras says it's down to the sensor's use of 'metamaterials' on its surface, which magnify the penetrating signals to give more workable results.
"We've seen so many people fail to crack this problem that many have become sceptical," he adds. "We were told to do an animal trial to get data – on pigs, as they're very resistant to insulin – and the results were fantastic. We were able to record results at some of the lowest concentrations ever captured.
"We have a plan, we are definitely on a good path. We want to work with a large OEM, because if successful this might change the industry."
Palikaras also very charitably suggested we read a continually updated PDF authored by lifelong chemist and noninvasive glucose testing expert John L Smith, a man who spent a vast chunk of his career appraising such tech on behalf of his pharma bosses.
We say charitably because Smith's book The Pursuit of Noninvasive Glucose: "Hunting the Deceitful Turkey" compares GlucoWise's metamaterials technology to the stuff of Harry Potter, also going to great lengths to detail the hundreds of failed attempts to produce a noninvasive monitor over the past three decades.
It's so negative, so littered with tales of people purposefully pursuing dead ends to bag investment cash, and with details of the technical problems faced in checking blood glucose through the skin, that you can't help but be grateful the simple blood test exists.
And you really don't want to know what Smith thinks about Google's glucose-sensing contact lens. [Spoiler alert: he says no one has ever proven a link between eye fluid glucose and actual blood levels, so it appears to be almost literally a complete fabrication. Or at best, some guesswork.]
Beating sugar
The other exciting new tech thing on the glucose-testing scene at the moment is the SugarBEAT, or at least the "coming late 2016" claims of its developer, Nemaura Medical.
This ought to be a more doable product than the sci-fi scanning of the GlucoWise, as SugarBEAT uses a known approach to testing that's worked with some small success in the past. It passes an electrical current over the skin that, Nemaura says, draws out "selected molecules" into its wearable sensor.
Bashir Timol, Nemaura Medical's director of business strategy, told us of his team's complex mission: "Challenges to date have included physics, electrochemistry, mechanical engineering, software and algorithm development," which is a nice summary of why glucose testing is such a difficult thing to do – and why your phone can't just do it by shining an LED at your wrist.
We asked Timol why, like many diabetes cures and treatments, non- or minimally-invasive testing always seems to be frustratingly out of reach, and whether there's any real hope on the horizon. He told us: "Whilst non-invasive systems have been in development for more than 20 years, it is recent advancements in nanotech that have the potential to revolutionise the diagnostic market."
He added: "We expect diagnostics may converge with wearable tech given the widespread adoption of smart watches/phones, which makes it much easier to have a patch-centric approach linked to an app."
SugarBEAT says it expects to launch at the end of 2016, with its system using one disposable patch per day, and saving on cost by offering the option of a phone and app to handle the data sent from a small Bluetooth dongle, meaning only the patch needs to be replaced.
John L Smith gives this idea a rough ride in his thorough appraisal though, pointing out that sucking things through the skin isn't necessarily proof or correlation of what's under there, and in what quantities.
Proving a correlation between the stuff that's drawn out of the skin and what lies beneath the skin – especially given the tiny quantities in which glucose is measured – is the big problem.
Simon O'Neill from Diabetes UK agrees, saying of such approaches: "The tech works, but whether the results are clinically accurate is another thing."
Encapsulating the problem
At what could be considered the fantasy/sci-fi end of the spectrum is biotech firm Viacyte. It's aiming big, and wants to effectively solve the problem of diabetes altogether by encapsulating insulin-producing cells within a microscopic structure.
Because Type 1 diabetes is an autoimmune disease, transplants aren't the answer: put a fully functioning pancreas into someone with Type 1 diabetes and the body's immune system instantly starts attacking it again, unless the recipient takes immunosuppressant drugs.
To counter this, Viacyte encapsulates the insulin-producing beta cells to stop the body's defences getting at them. There's another problem, though: the cells need to breathe to stay alive, so the protective structure has to be smart enough to block immune attacks while also allowing oxygen and blood vessels in.
And that's where it gets complicated. Viacyte is actually combining two products into its VC-01: the PEC-01 system of creating the beta cell precursors that eventually mature into insulin producing islet cells, and the Encaptra pod that protects the implanted cells from the immune attack once they're inside the body.
That might mean we're looking at not one but two products that never quite work as advertised, and which eventually slide into development limbo and never see the light of day.
However, Diabetes UK's Simon O'Neill thinks this approach is likely to be the one that will one day free diabetes sufferers from blood tests – and hopefully makes noninvasive sensor tech obsolete before anyone even proves it works.
"I think we might be getting to a stage where a cure is 10 years away, or maybe less," O'Neill said. "Something like protected islet cell implants that are topped up in a hospital every six months would not be a permanent cure, but may well be something which is going to happen."
So will any of this happen?
The one thing everyone we talked to continually stressed is that we're still in the early stages of development of noninvasive testing. Which is frustrating, because 10 years ago quite similar – although less well promoted – technologies were also in the early stages of development, but were either proven impractical, or simply didn't work.
Indeed, as we were researching this feature another announcement was made by UK researchers at Cardiff University's School of Engineering, who claim to have a new concept for a microwave device that may also deliver the noninvasive, through-the-skin testing of diabetes sufferer's dreams.
However, they admit to being five years away from market – which is at least a bit better than ten – and in need of funding.
You'd think that if such a gadget was to ever reach the home market there would already be a car-sized machine doing the job in hospitals, but there isn't. It's not just a question of miniaturisation, it's about inventing a completely new type of medical technology – one that, while easy to promise on paper, could remain unrealised for generations to come.
And the underlying message from those working in the field seems to be: "We're trying, but don't get your hopes up."